Assess Parkinson’s symptoms independently and consistently
The sensitivity and variability in measurement are critical for clinical trials. Ensuring sensitivity in your measurement tool can ensure that you’re not missing important features. Minimizing variability by improving test-retest reliability can improve the power of your study.
Monitor Motor Symptom Changes with Sensitivity
In terms of Kinesia technology, ‘sensitivity’ refers to important measurement issues that impact clinical trial outcomes: Measurements such as Symptom Differentiation and Minimal Detectable Change
Symptom Differentiation
Different treatment protocols may have a unique impact on different symptoms.
A treatment that improves one symptom, like tremor, may not have an impact on another – say bradykinesia. What’s more, different treatments impact different features of a particular symptom. In a similar fashion, one treatment strategy may improve bradykinesia speed but have no effect on bradykinesia rhythm.
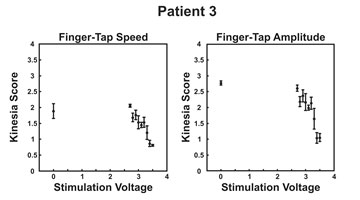
For the patient represented in Figures 1 and 2 above, treatment with deep brain stimulation had little to no impact on tremor yet provided fine, graded control of finger tapping speed and amplitude associated with bradykinesia. This emphasizes that if there is no differentiation of impact on specific symptoms, important results can be overlooked.
Minimal Detectable Change (MDC)
The smallest significant difference that can be detected with a measurement tool.
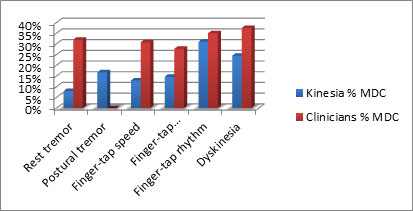
Smaller minimal detectable change means fewer data points are required to demonstrate a significant effect. A recent clinical study has demonstrated that Kinesia technology provides a smaller minimal detectable change when compared to traditional rating scales. This is particularly important for studies evaluating neuroprotective effects.
As a percent of full measurement scale, Kinesia technology allows detection of smaller minimal detectable changes across symptoms.
Minimize the Error Bars (Variability)
Traditional rating scales are subject to bias which can impact both intra- and inter-rater reliability of measurements
Test-Retest reliability is the output repeatability of a measurement tool for the same input. For example, if the same level of dyskinesia is present in a specific patient at different times, will the measurement tool provide the same output? Similarly, if two different patients have the same level of dyskinesia, will the measurement tool provide the same result for both patients?
Intra-rater reliability
The repeatability of a single clinician to rate the same symptom severity at different points in time.
Inter-rater reliability
The repeatability of multiple clinicians (which is often the case in clinical studies) to rate symptoms the same.
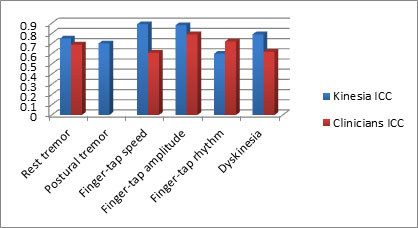
Figure 4
As calculated by the IntraClass Correlation, Kinesia technology
provides improved test-retest reliability compared to traditional
rating scales.
Kinesia technology provides an objective and unbiased tool to measure motor symptom severity. The test-retest reliability of a measurement tool is calculated by the IntraClass Correlation (ICC). A recent clinical study demonstrated that Kinesia technology provides a higher ICC for symptoms when compared to subjective rating scales. Improving the test-retest reliability of your measurement device decreases variability in your study.
Another traditional measurement tool is patient diaries. Patient diaries are greatly subject to patient compliance, recall, and accuracy. The accuracy of patient diaries can be degraded both by a patient’s ability to differentiate motor symptoms, such as tremor from dyskinesia, as well as recall: Patients may not complete their diaries at specified times and then face difficulty remembering symptom states for a particular point in time. Kinesia’s HomeView system is equipped with alarms to remind patients to complete motor assessment tasks at specific times throughout the day. The system time-stamps each task to indicate when exactly the task was performed. The HomeView system’s alarm solves patient compliance issues and the video capture and electronic diary solve the recall and accuracy issues; solutions integral for minimizing variability for clinical trial data.